Credits/Names: Japan Science and Technology Agency, DMR-1420620 and DMR-1210588, Japan Society for the Promotion of Science for Research Abroad (No. 25-185), Spring-8 Synchrotron Source. Takeshi Yajima,1 Fumitaka Takeiri,1 Kohei Aidzu,1 Hirofumi Akamatsu,2 Koji Fujita,1 Masatoshi Ohkura,1 Wataru Yoshimune,1 Shiming Lei,2 Venkatraman Gopalan,2 Katsuhisa Tanaka,1 C. M. Brown,3 Mark A. Green,4 Takafumi Yamamoto, 1Yoji Kobayashi1 and Hiroshi Kageyama1; 1 Kyoto Univ., 2 Penn State, 3 NIST, 4 Univ. of Kent. Nature Chemistry, 7, 1017–1023 (2015).
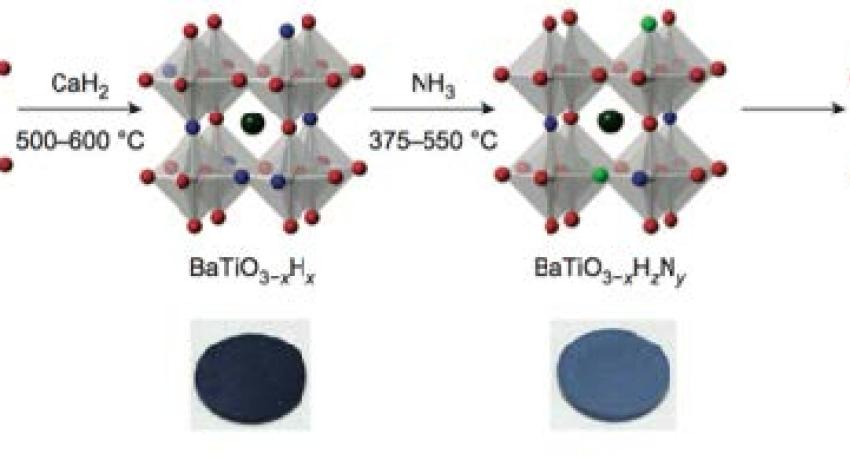
Oxynitrides are attractive due to a combination of visible-light absorption, photocatalytic activity, and high dielectric permittivity. Their synthesis typically requires high-temperature NH3 treatment of oxides, but the highly reducing conditions and the low mobility of N3– greatly constraint the composition, structure, and hence properties of the resulting oxynitrides. A MRSEC team has demonstrated a topochemical route to making oxynitrides at less than 500°C using a perovskite oxyhydride as a host, obtaining a room-temperature ferroelectric BaTiO3–xN2x/3. Anion exchange is accompanied by a metal-to-insulator cross-over via mixed O–H–N intermediates. This “labile hydride” strategy can now be applied to other oxynitrides and perhaps other mixed anionic compounds. BaTiO3–xN2x/3 also has potential applications in next-generation electronics.
Credits/Names: Japan Science and Technology Agency, DMR-1420620 and DMR-1210588, Japan Society for the Promotion of Science for Research Abroad (No. 25-185), Spring-8 Synchrotron Source. Takeshi Yajima,1 Fumitaka Takeiri,1 Kohei Aidzu,1 Hirofumi Akamatsu,2 Koji Fujita,1 Masatoshi Ohkura,1 Wataru Yoshimune,1 Shiming Lei,2 Venkatraman Gopalan,2 Katsuhisa Tanaka,1 C. M. Brown,3 Mark A. Green,4 Takafumi Yamamoto, 1Yoji Kobayashi1 and Hiroshi Kageyama1; 1 Kyoto Univ., 2 Penn State, 3 NIST, 4 Univ. of Kent. Nature Chemistry, 7, 1017–1023 (2015).
Download PDF Version: BaTiON.pdf
Year of Highlight: 2016
IRG: IRG 1 - Designing Functionality into Layered Ferroics